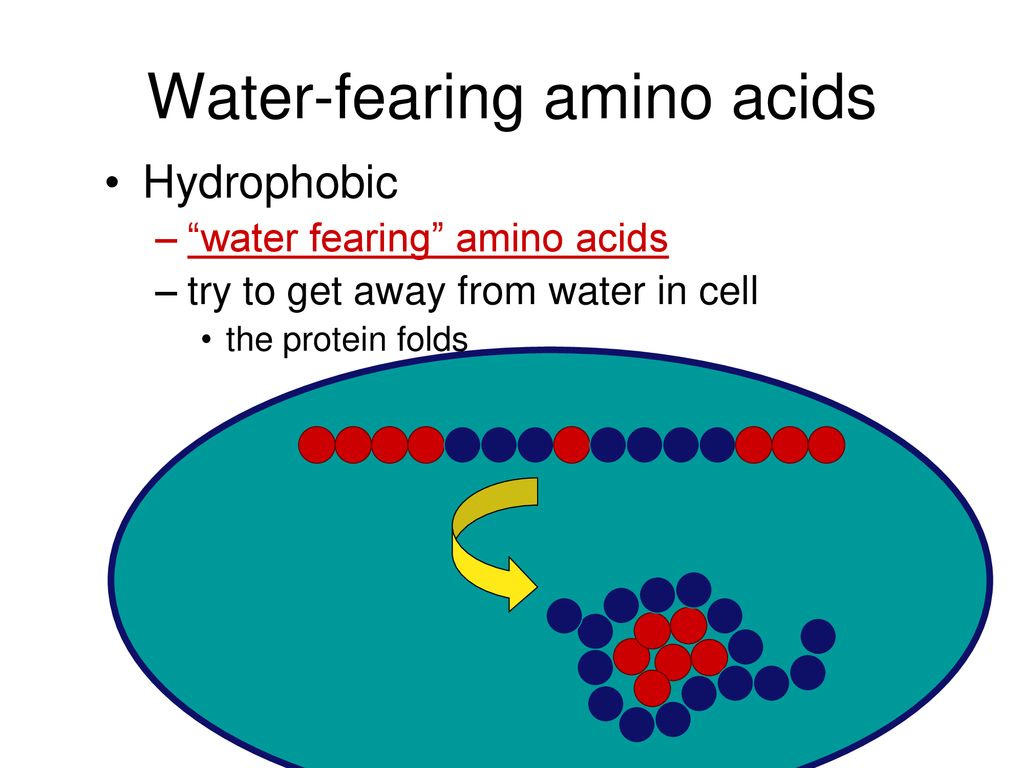
Inside this core the hydrophobic side chains are shielded from the water. When a protein folds, hydrophobic amino acids get buried inside the protein to form a hydrophobic core. For example, the design of enzymes that are stable and functional at low temperatures may benefit from this work. Understanding the temperature dependence for amino acids, can help to make proteins (or enzymes) stable at a specific temperature range. This approach shows that the hydrophobic effect becomes weaker at lower temperatures, as expected from theoretical predictions. For each amino acid type, we use the ratio between the number of residues at the inside and at the surface of the folded structures as a measure for its hydrophobicity. Here we are able to estimate the strength of the hydrophobic effect, by analysing the positions of a large number of amino acids from protein structures experimentally determined at different temperatures. Nevertheless, it is difficult to quantify the temperature dependence for hydrophobic amino acids.
However, the strength of the hydrophobicity is known to be strongly temperature dependent, leading for example to lower stability at lower temperatures (cold denaturation). On folding, hydrophobic amino acids get buried inside the protein such that they are shielded from the water this hydrophobic effect makes a protein fold stable. In general, proteins become functional once they fold into a specific globular structure. Moreover, this work provides a method for probing the individual temperature dependence of the different amino acid types, which is difficult to obtain by direct experiment. Alternatively, one can conclude that the temperature dependence of the hydrophobic effect has a measurable influence on protein structures. These propensities show that the hydrophobicity becomes weaker at lower temperatures, in line with current theory. Using NMR structures filtered for sequence identity, we were able to extract hydrophobicity propensities for all amino acids at five different temperature ranges (spanning 265-340 K). Here we investigate if it is possible to extract this temperature dependence directly from a large set of protein structures determined at different temperatures. This temperature dependence is thought to explain the denaturation of proteins at low temperatures. However, the hydrophobic force is known to be strongly temperature dependent. One can estimate the relative strength of this hydrophobic effect for each amino acid by mining a large set of experimentally determined protein structures. The hydrophobic effect is the main driving force in protein folding.
